100205
论文已发表
提 交 论 文
注册即可获取Ebpay生命的最新动态
注 册
IF 收录期刊
- 3.3 Breast Cancer (Dove Med Press)
- 3.4 Clin Epidemiol
- 2.5 Cancer Manag Res
- 2.9 Infect Drug Resist
- 3.5 Clin Interv Aging
- 4.7 Drug Des Dev Ther
- 2.7 Int J Chronic Obstr
- 6.6 Int J Nanomed
- 2.5 Int J Women's Health
- 2.5 Neuropsych Dis Treat
- 2.7 OncoTargets Ther
- 2.0 Patient Prefer Adher
- 2.3 Ther Clin Risk Manag
- 2.5 J Pain Res
- 2.8 Diabet Metab Synd Ob
- 2.8 Psychol Res Behav Ma
- 3.0 Nat Sci Sleep
- 1.8 Pharmgenomics Pers Med
- 2.7 Risk Manag Healthc Policy
- 4.2 J Inflamm Res
- 2.1 Int J Gen Med
- 4.2 J Hepatocell Carcinoma
- 3.7 J Asthma Allergy
- 1.9 Clin Cosmet Investig Dermatol
- 2.7 J Multidiscip Healthc

已发表论文
替米考星氢化蓖麻油纳米颗粒的不同颗粒大小悬浮液的制备与评价
Authors Chen X, Wang T, Lu M, Zhu L, Wang Y, Zhou W
Published Date May 2014 Volume 2014:9(1) Pages 2655—2664
DOI http://dx.doi.org/10.2147/IJN.S58898
Received 8 December 2013, Accepted 22 January 2014, Published 27 May 2014
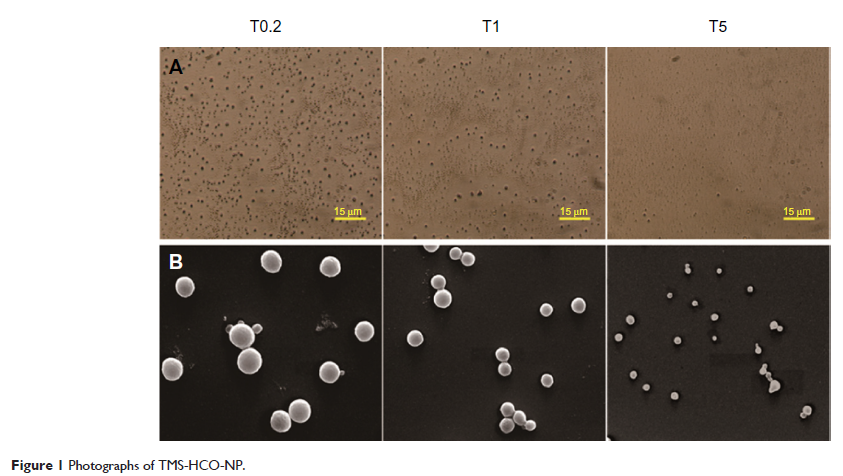
Abstract: Three tilmicosin-loaded hydrogenated castor oil nanoparticle (TMS-HCO-NP) suspensions of different particle sizes were prepared with different polyvinyl alcohol surfactant concentrations using a hot homogenization and ultrasonic technique. The in vitro release, in vitro antibacterial activity, mammalian cytotoxicity, acute toxicity in mice, and stability study were conducted to evaluate the characteristics of the suspensions. The in vitro tilmicosin release rate, antibacterial activity, mammalian cytotoxicity, acute toxicity in mice, and stability of the suspensions were evaluated. When prepared with polyvinyl alcohol concentrations of 0.2%, 1%, and 5%, the mean diameters of the nanoparticles in the three suspensions were 920±35 nm, 452±10 nm, and 151±4 nm, respectively. The three suspensions displayed biphasic release profiles similar to that of freeze-dried TMS-HCO-NP powders, with the exception of having a faster initial release. Moreover, suspensions of smaller-sized particles showed faster initial release, and lower minimum inhibitory concentrations and minimum bactericidal concentrations. Time-kill curves showed that within 12 hours, the suspension with the 151 nm particles had the most potent bactericidal activity, but later, the suspensions with larger-sized particles showed increased antibacterial activity. None of the three suspensions were cytotoxic at clinical dosage levels. At higher drug concentrations, all three suspensions showed similar concentration-dependent cytotoxicity. The suspension with the smallest-sized particle showed significantly more acute toxicity in mice, perhaps due to faster drug release. All three suspensions exhibited good stability at 4°C and at room temperature for at least 6 months. These results demonstrate that TMS-HCO-NP suspensions can be a promising formulation for tilmicosin, and that nanoparticle size can be an important consideration for formulation development.
Keywords: tilmicosin, solid lipid nanoparticle suspension, particle size, release rate, antibacterial activity, toxicity
Keywords: tilmicosin, solid lipid nanoparticle suspension, particle size, release rate, antibacterial activity, toxicity
Download Article[PDF]